How our plastic fragments are made
A proprietary, patent-pending workflow designed to generate realistic micro- and nanoplastic fragments
1. We start with real-world plastic feedstocks
Industrial-grade materials used in plastic manufacturing
More representative than narrowly defined lab polymers
Verified by FTIR and GEC for identity, additive analysis, and consistency

2. We mechanically break plastics down without heat damage
Cryogenic grinding and low-temperature milling (<15 °C)
Preserves native polymer structure without thermal deformation
Generates irregular, non-spherical fragments
Size-controlled, polydisperse populations (~100 nm, 1 µm, 10 µm)

fragments centered ~100nm (TEM)

Target size ranges defined within controlled windows
Dispersant removed and replaced with minimal BSA stabilization
Maintains dispersion while preserving native surface behavior
3. We define size and stabilize for biological use
size enrichment
BSA-coronated fragments (TEM)

4. We validate performance in real biological systems
Morphology and size characterized by TEM, LD, MADLS, and NTA
Fragment stability evaluated in biologically relevant solutions
Cellular responses assessed by microscopy and imaging flow cytometry
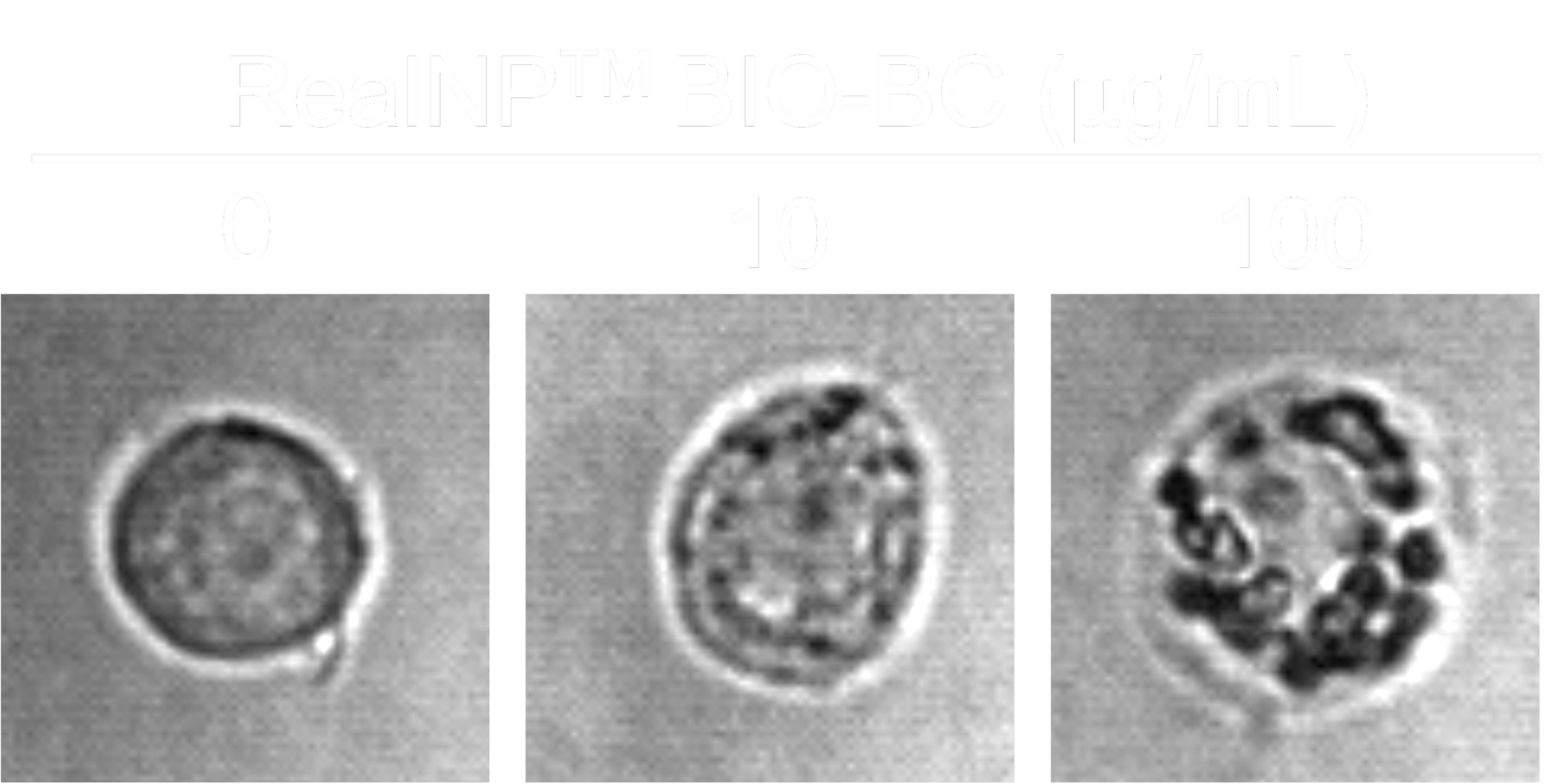
cell responses upon nanoplastic exposure
RAW264.7 cells after 24h
Our product lines
RealNP™-CORE
Mechanically generated 100 nm range polystyrene nanoplastic fragments
Surfactant-stabilized to maintain dispersion without aggregation
No antimicrobial preservatives (e.g., sodium azide), minimizing unintended toxicity artifacts
Optimized for detergent-insensitive analytical method development
Enables direct comparison with prior nanosphere-based literature
Mechanically generated polystyrene nanoplastic fragments
Surfactant-stabilized to maintain dispersion without aggregation
No antimicrobial preservatives (e.g., sodium azide), minimizing unintended toxicity artifacts
Optimized for detergent-insensitive analytical method development
Enables direct comparison with prior nanosphere-based literature
RealNP™-BIO-BC
Mechanically generated 100nm range polystyrene nanoplastic fragments
Minimally BSA-coated surface with near-complete removal of detergent (<0.000005% w/v)
Maintains stable dispersion across biologically relevant solutions (plasma, media, buffers)
Optimized for high-sensitivity analytical methods sensitive to salts and surfactants (e.g., MS)
Reduces confounding variables while preserving native particle–protein interactions
Mechanically generated polystyrene nanoplastic fragments
Minimally BSA-coated surface with near-complete removal of detergent (<0.000005% w/v)
Maintains stable dispersion across biologically relevant solutions (plasma, media, buffers)
Optimized for high-sensitivity analytical methods sensitive to salts and surfactants (e.g., MS)
Reduces confounding variables while preserving native particle–protein interactions
We offer 3 different sized units designed for:
1 mg — Detection method development (ships immediately)
Ideal for calibration, sensitivity testing, and early-stage assay development5 mg — In vitro studies (available week of April 27)
Suitable for cell-based assays, uptake studies, and iterative experimental work20 mg — Extended and in vivo studies (available week of April 27, contact us before ordering)
Designed for larger-scale experiments, longitudinal studies, and in vivo applications
Frequently Asked Questions
Answers to common questions about our materials, applications, and best practices. Can’t find your answer? Please reach out to us!
-
Yes. We currently accept credit card payments and are expanding to support:
Purchase Orders (POs)
Invoice-based payments (e.g., Net terms)
Integration with research procurement platforms (e.g., Quartzy)
Procurement through scientific distributors (e.g., Fisher Scientific, VWR)
If your institution requires a specific payment method, please contact us — we’re happy to work with your procurement team.
-
We are expanding across several dimensions:
Polymer types: PET, PE, PP, and other common plastics
Size ranges: extending beyond current ~100 nm, 1 µm, and 10 µm populations
Weathering states: oxidation, UV exposure, and environmentally aged materials
Surface formats: controlled protein corona systems and ligand-functionalized particles
Labeling options: fluorescence-labeled fragments for uptake and tracking studies
-
BSA is used to stabilize particle dispersion while introducing minimal additional variables.
Biologically familiar: Cell culture media typically contains serum, where albumin is the dominant protein
Low reactivity: Albumin is structurally robust and among the least reactive proteins
Minimal interference: Provides a neutral surface without imposing strong or artificial functionality
Controlled baseline: Establishes a simple, reproducible starting surface for experiments
Biologically relevant starting point: Albumin is among the first proteins to adsorb to particle surfaces in biological fluids, and BSA coating captures this initial step in a controlled way
In biological environments, the initial BSA layer is partially replaced by surrounding proteins, allowing the particle surface to adapt to the experimental context.
-
Dose is highly dependent on cell type and experimental context.
As a general guideline:
10–500 µg/mL is a practical working range for most in vitro studies
≥1000 µg/mL is typically not biologically relevant and may introduce artifacts due to particle crowding and aggregation
We recommend starting low and titrating based on your specific model.
-
Cellular sensitivity is largely driven by uptake behavior, not inherent tolerance.
Macrophage-like cells (e.g., RAW264.7)
Highly phagocytic
Uptake readily → responses observed at ~10 µg/mL
Structural cells (e.g., epithelial cells)
More selective uptake
Lower apparent sensitivity unless particles are internalized
This difference reflects cellular selectivity, not resistance.
-
Particles can be modified to enhance uptake, for example through:
Ligand-mediated targeting
Protein corona engineering
Protocols for controlled surface functionalization will be made available.


